Significant Achievement! Mednovo Group's Self-Developed Microwave Ablation System Publishes Important Research Findings in Top International Journal
Recently, multiple scholars from six domestic medical centers published a significant article titled "Safety and Efficacy of Microwave Versus Laser Ablation in the Treatment of Varicose Veins of Lower Extremities (MOST Trial): A Multicenter, Open-Label, Non-Inferiority, Randomized Controlled Trial" in the International Journal of Surgery.
Source

[International Journal of Surgery]
10.1097/JS9.0000000000003941
Published: November 13, 2025 Impact Factor: 10.3
This study aimed to evaluate the non-inferiority of two ablation techniques in treating great saphenous vein varicosities, providing important guidance for selecting appropriate treatment methods in clinical practice.
Authors

About Lower Extremity Varicose Veins and Treatment Methods
Lower extremity varicose veins are a type of chronic venous disease and one of the most common conditions in vascular surgery. The core mechanism involves venous dysfunction and valvular reflux caused by obstructed blood return, leading to increased venous pressure, superficial vein prominence, and tortuous dilation. Approximately 25% of adults worldwide are affected, with prevalence rising in China due to an aging population and lifestyle changes. According to the International Union of Phlebology, the incidence of chronic venous disease in individuals aged around 50 with lower extremity discomfort is 63.9% globally.
Traditional treatments for varicose veins have various limitations. Compression therapy and medication, such as elastic stockings or diosmin, are suitable for early-stage patients to alleviate symptoms but cannot provide a cure. Open surgeries, like high ligation and stripping of the great saphenous vein, require incisions in the skin and subcutaneous tissues, followed by cutting, ligation, and removal of diseased veins. These procedures involve significant trauma, slow recovery, and considerable patient discomfort. Consequently, minimally invasive treatments have become essential. Techniques like endovenous laser ablation (EVLA), often combined with foam sclerotherapy, radiofrequency ablation (RFA), and microwave ablation (MWA), represent the current trend in treating lower extremity varicose veins.
The article notes that traditional surgical treatments, such as high ligation and stripping, have largely been replaced by endovenous thermal ablation (ETA). Compared to endovenous laser ablation (EVLA) and radiofrequency ablation (RFA), endovenous microwave ablation (EMA)ЁЊa novel ETA methodЁЊgenerates heat through microwave radiation, causing high-frequency vibration of polar molecules within the venous wall. This enables more uniform tissue heating over a broader area at lower temperatures, reducing thermal damage to tissues.Research Findings
1. Non-Inferior Efficacy
Microwave ablation was non-inferior to laser ablation in the primary efficacy endpoint (great saphenous vein occlusion rate) at 6 months post-operation. Long-term follow-up showed stable outcomes, with high cumulative closure rates maintained at 12 months (EMA 90.41% vs. EVLA 84.21%).
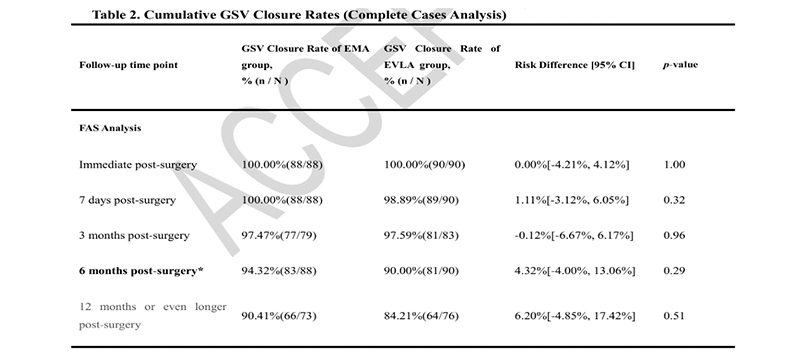
The study met its predefined primary efficacy endpoint. EMA was non-inferior to EVLA in vessel closure rates at 6 months post-operation.
The lower limit of the 95% CI for the risk difference was > -10%, with a non-inferiority test P-value < 0.001.
2. Significant Improvement in Quality of Life
Both groups showed significant reductions in Venous Clinical Severity Score (VCSS) and Aberdeen Varicose Vein Questionnaire (AVVQ) scores within 12 months post-operation, indicating marked symptom improvement.
No significant differences were observed between groups in quality-of-life improvements, suggesting EMA is comparable to EVLA in symptom relief
3. Excellent Safety Profile
No significant differences were found in overall adverse event rates between groups (EMA 37.50% vs. EVLA 42.22%).
No device-related serious adverse events or device defects were reported.
Microwave ablation operates at lower temperatures (70ЈC100ЁуC vs. laser >100ЁуC and radiofrequency at 120ЁуC), theoretically reducing thermal damage and postoperative pain.
Conclusions from the Trial
ЁЄ EMA serves as an effective alternative to EVLA, demonstrating similar or superior safety, efficacy, and operational efficiency. The findings support the feasibility of EMA as a first-line treatment option in clinical practice.
ЁЄ This study is the first multicenter randomized controlled trial (RCT) to provide high-quality evidence for the global promotion and application of microwave ablation technology.
ЁЄ This study was supported with equipment and funding by Beijing Sanhe Dingye Technology Co., Ltd., a subsidiary of Mednovo Group.
ЁЄ The research adhered to the CONSORT 2025 and TITAN 2025 guidelines, with transparent and publicly available data for academic exchange.

The UM200 Microwave Ablation System (Registration Certificate No.: National Medical Device Approval 20233011741) and its supporting novel water-cooled disposable microwave ablation catheter (Registration Certificate No.: National Medical Device Approval 20253012201), designed and developed by Beijing Sanhe Dingye Technology Co., Ltd., a subsidiary of Mednovo Group, have successfully obtained Class III medical device registration approval from the National Medical Products Administration (NMPA) and are now commercially available. This innovation provides a safe, efficient, and minimally invasive treatment option for great saphenous vein varicosities, perfectly aligning with the modern healthcare principle of "patient-centered" care and becoming the preferred choice for both clinicians and patients.
The system achieves precise treatment through a thermal ablation mechanism. Under ultrasound guidance, it enables a zero-incision, non-surgical procedure by inserting the disposable microwave ablation catheter into the target vessel. The microwave ablation system delivers high-precision microwave energy, combined with linear control technology, to ensure continuous microwave output with a frequency of 2450ЁР50Hz, a power range of 0ЈC70W, and a step increment of 1W, offering accurate frequency control and high thermal efficiency.
Initially, the product design did not incorporate temperature monitoring or cooling mechanisms, requiring the use of wet gauze to wrap the catheter during procedures. In response to clinical challenges and feedback, the R&D team at Mednovo Group integrated an all-in-one water-cooling circulation system and a temperature monitoring system into the product. This effectively reduces catheter body temperature, monitors ablation zone and catheter temperatures in real-time, and prevents scalding injuries to both patients and medical staff. Through bench testing and ex vivo studies, the product performance was further optimized, and patents for the microwave therapy device and microwave ablation catheter were granted (Patent No.: ZL20210733837.2). This achievement is the result of collaborative efforts between Professor Weiwei Wu from Beijing Tsinghua Changgung Hospital, Tsinghua University, and the R&D team at Mednovo Group, exemplifying successful in-depth collaboration between medicine and industry, interdisciplinary integration of medicine and engineering, and efficient translation of research into practical solutions.

In November 2025, the second-generation disposable microwave ablation catheter was approved for market launch, with the registration certificate number: Guo Xie Zhun 20253012201. The new-generation microwave ablation catheter features a reduced diameter of 6F (2mm), minimizing surgical trauma and enhancing patient comfort. Additionally, precise marking indicators have been added to the catheter to assist physicians in accurate intraoperative positioning, thereby improving surgical precision. Leveraging its independent research and development capabilities, Beijing Sanhe Dingye Technology has achieved domestic innovation, ensured independent controllability, and possesses full intellectual property rights, ultimately benefiting a broad range of patients.
